Global Post-Market Compliance Services for Regulated Devices
Global post-market compliance requires more than documentation. It demands a partner who can connect field data with risk management, product improvement, regulatory reporting, and lifecycle decisions. Syrma Johari MedTech delivers end-to-end post-market compliance support for medical devices and IVDs, helping manufacturers strengthen patient safety, maintain market access, and respond faster to evolving global regulations.
Beyond Surveillance. Full Post-Market Compliance.
Post-market compliance includes all regulatory and quality activities required after a device or IVD enters the market. It extends beyond surveillance alone to cover complaint handling, reportable events, vigilance submissions, field safety corrective actions, recalls, PMCF for devices, PMPF for IVDs, periodic safety reporting, and continuous benefit-risk evaluation within the quality management system.
Post-Market Surveillance
- Ongoing collection and evaluation of real-world device and IVD performance data.
Complaint Handling & Vigilance
- Complaint intake, triage, investigation, reportability assessment, and authority submissions.
PMCF / PMPF
- Post-market clinical and performance follow-up programs to confirm safety, performance, and emerging risk trends.
Recalls, FSCA & CAPA
- Corrective field actions, recall management, root cause investigation, and CAPA closure.
Why Manufacturers Need an Integrated Post-Market Model
Regulators increasingly expect proactive, connected post-market systems. Reactive complaint handling, fragmented vendors, and disconnected quality processes create risk across audits, reporting timelines, and market continuity. When post-market data do not flow into design controls, risk files, CAPA, and manufacturing changes, compliance gaps become harder to close.
Reactive systems create audit risk
EU MDR, IVDR, FDA, and other regulators require structured, proactive post-market programs.
Global reporting is complex
Each market has different reporting rules, timelines, portals, and submission expectations.
Disconnected teams slow action
When quality, engineering, manufacturing, and regulatory partners are siloed, field signals do not convert quickly into corrective action.
Lifecycle feedback is essential
Strong post-market systems improve risk management, labeling, training, design decisions, and product reliability.
Post-Market Compliance Connected to Design, Manufacturing, and Quality
Post-market compliance becomes more effective when the same partner who supports design, manufacturing, testing, and regulatory functions can also manage complaints, vigilance, PMCF/PMPF, and regulatory sustenance. This creates a closed-loop model where field feedback drives faster root cause analysis, more effective CAPA, better design updates, and stronger regulatory closure.
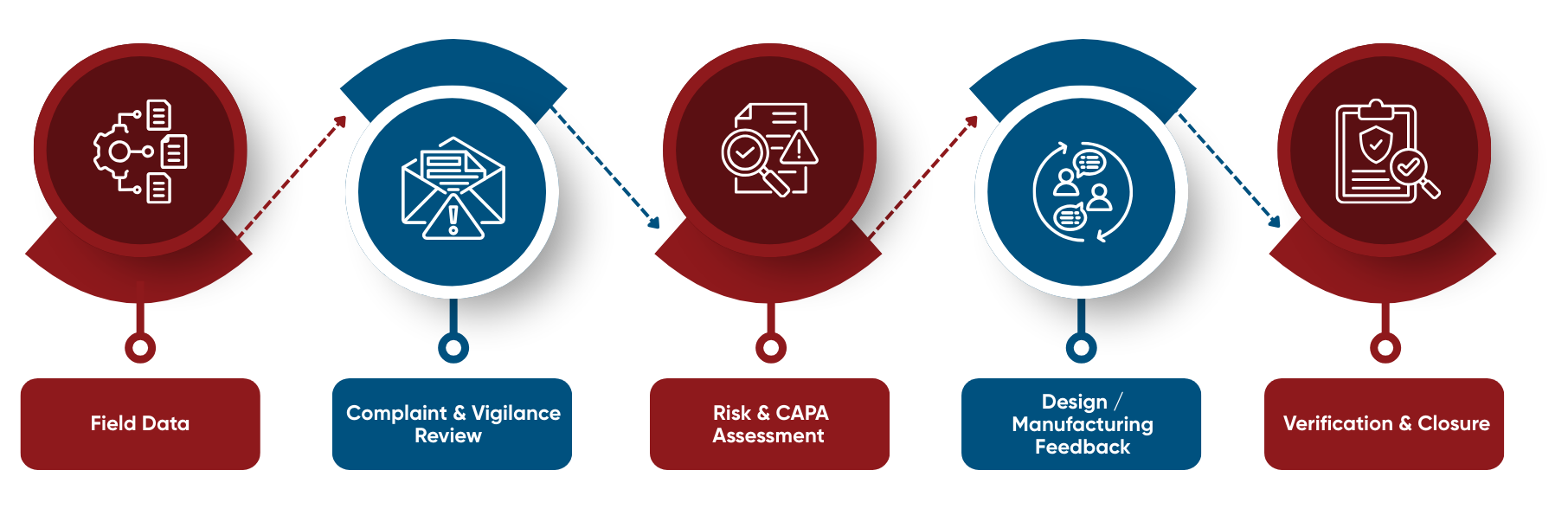
Our End-to-End Post-Market Compliance Framework
Aligned with ISO 13485:2016, ISO 14971:2019, and ISO/TR 20416:2020
Ensure your PMS plan, complaint handling processes, vigilance reporting, and PSUR documentation align with EU MDR requirements. This toolkit provides practical checklists and templates used by regulatory teams to implement MDR-compliant post-market surveillance programs.
Strategy & Governance
Global PMS frameworks aligned to EU MDR/IVDR, FDA, TGA, Health Canada, MHRA, PMDA, NMPA, CDSCO, and other major regulatory systems. We define governance structures, responsibilities, escalation pathways, and device-family PMS plans.
PMS Planning & Documentation
Development of PMS plans, PMS reports, PSURs, data aggregation strategies, notified body-ready dossiers, and authority submission support for medical devices and IVDs.
PMCF & PMPF Programs
PMCF strategy, PMPF planning, protocol development, literature-based follow-up, registry participation, and integration of findings into CERs and PERs.
Complaint Handling & Vigilance
Complaint intake, triage, investigation, trend analysis, signal detection, reportability determination, and jurisdiction-specific adverse event submissions across key markets.
FSCA, Recalls & CAPA
Health hazard evaluation, recall strategy, FSN drafting, field action execution, effectiveness checks, root cause analysis, CAPA implementation, and closure documentation.
Regulatory Intelligence & Remediation
PMS gap assessments, inspection readiness support, remediation of 483s and non-conformities, and ongoing monitoring of evolving global guidance and regulations.
Data Management & Analytics
Complaint and vigilance databases, ERP/eQMS/LIMS/CRM integration, KPI dashboards, automated workflows, and traceable documentation for audits and inspections.
Why Syrma Johari for Post-Market Compliance
Integrated Lifecycle Partner
We connect post-market compliance with design, engineering, manufacturing, testing, and regulatory sustenance.
Global Regulatory Reach
Support across EU, US, UK, Canada, Australia, Japan, China, India, Brazil, and Gulf markets.
Medical Device & IVD Expertise
Experience across diagnostics, monitoring, drug delivery, therapy, critical care, and complex regulated product categories.
MDSAP-Certified Quality Foundation
Quality systems aligned with global regulatory expectations, supported by SJML’s certified infrastructure.
Scaled Execution Capability
14 manufacturing facilities and 4 design centers supporting regulatory and lifecycle needs at scale.
International Registration Experience
Regulatory approvals across 50+ countries, including US FDA 510(k) clearances and CDSCO licenses, strengthening post-market sustenance capability.
How We Build and Run Post-Market Compliance Programs
Step 1 — Discovery & Gap Assessment
We assess existing PMS systems, complaint handling, vigilance workflows, periodic reports, CAPA records, and regulatory documentation against target-market requirements.
Deliverables:
Gap assessment, risk prioritization, remediation roadmap
Step 2 — Strategy & Planning
We design the post-market compliance framework around your portfolio, markets, and internal operating model, including PMS plans, governance, reporting logic, and follow-up strategy.
Deliverables:
Global framework, device-family PMS plans, SOP recommendations, PMCF/PMPF strategy
Step 3 — Implementation & Operationalization
We establish workflows, systems, reporting mechanisms, escalation paths, and training programs. For outsourced models, we can operate the process with agreed SLAs.
Deliverables:
Operational workflows, trained teams, validated processes, submission-ready systems
Step 4 — Continuous Compliance & Sustenance
We provide ongoing support for PMSR, PSUR, PMCF/PMPF execution, vigilance reporting, recalls, trend analysis, and regulatory intelligence.
Deliverables:
Periodic reports, vigilance submissions, trend reviews, regulatory updates, continuous improvement support
Flexible Engagement Models for Different Lifecycle Needs
Project-Based Support
Ideal for defined deliverables such as PMS plans, PSURs, PMCF/PMPF protocols, gap remediation, or technical documentation updates.
Managed Service / FSP Models
Outsourced complaint handling, vigilance reporting, trend analysis, recall coordination, and periodic reporting with end-to-end responsibility.
Hybrid Lifecycle Support
Integrated programs combining post-market compliance with design changes, verification, testing, manufacturing updates, and regulatory sustenance.
Representative Post-Market Compliance Outcomes
Our post-market compliance support helps clients strengthen reporting readiness, close regulatory gaps, and improve lifecycle responsiveness across global markets.
EU MDR Transition for Multi-Country Portfolio
Supported device-family PMS plans, PSUR authoring, and PMCF program setup for an EU device portfolio, helping achieve compliance with zero PMS-related notified body non-conformities.
FDA MDR & Recall Remediation for IVD Platform
Redesigned complaint handling and MDR workflows after FDA observations, improving on-time reporting and reducing CAPA cycle time.
Global Vigilance Outsourcing for Diagnostic Portfolio
Managed multi-country vigilance reporting and complaint operations, enabling rapid portfolio scaling without expanding internal headcount.
FSCA Management for Therapeutic Device
Supported hazard evaluation, field communication, authority notification, and CAPA execution for a multi-country field action with strong closure outcomes.
FSCA, Recalls & CAPA
Health hazard evaluation, recall strategy, FSN drafting, field action execution, effectiveness checks, root cause analysis, CAPA implementation, and closure documentation.
What Effective Post-Market Compliance Delivers
Reduced Recall Risk
- Earlier signal detection and more structured CAPA reduce escalation into major field actions.
Faster Regulatory Closure
- Improved reporting discipline and authority communication support faster issue closure.
Better Audit Readiness
- Traceable systems and stronger QMS integration reduce PMS-related audit observations.
Faster Corrective Implementation
- Closed-loop links to design and verification teams accelerate product and labeling changes.
Strengthen Post-Market Compliance Across the Full Device Lifecycle
Whether you are preparing for EU MDR or IVDR, improving complaint and vigilance systems, responding to audit findings, or scaling global reporting operations, Syrma Johari MedTech brings the regulatory, quality, engineering, and manufacturing expertise needed to build a more connected post-market system.
Information Center
What is the difference between post-market compliance and post-market surveillance?
Post-market surveillance is one part of post-market compliance. Post-market compliance is broader and includes PMS, complaint handling, vigilance, recalls, FSCA, CAPA, PMCF/PMPF, periodic safety reporting, and integration with risk management and design controls.
Which regulations drive post-market compliance for medical devices and IVDs?
Key frameworks include EU MDR, EU IVDR, FDA 21 CFR 803/806/820, ISO 13485, ISO 14971, and country-specific requirements in markets such as the UK, Canada, Australia, Japan, China, India, and Brazil.
How have EU MDR and IVDR increased post-market obligations?
EU MDR and IVDR require stronger PMS planning, PMSR/PSUR reporting, PMCF/PMPF, trend reporting, and deeper integration with clinical or performance evaluation, risk management, and EUDAMED-related documentation.
Can SJML support both new launches and commercial products?
Yes. We support pre-launch setup of PMS plans and complaint systems, ongoing post-market operations for commercial products, and sustenance programs for mature or legacy portfolios.
How does SJML connect post-market data with engineering and manufacturing?
Our integrated model links complaint and vigilance signals to engineering review, CAPA, verification, manufacturing change control, and product improvement activities for faster and more effective closure.
Do you work with start-ups as well as large OEMs?
Yes. Engagements can range from focused deliverables for emerging companies to global managed vigilance and hybrid lifecycle support models for large OEM portfolios.
COMPANY
- About Us
- Board Members & Leadership
- Certification and Awards
- ESG Initiatives
- Career
LEGAL
- CSR Policy
- Privacy Policy
- Terms & Conditions
CONTACT US
Business Inquiry
© 2026, Syrma Johari MedTech Ltd. All rights reserved.
