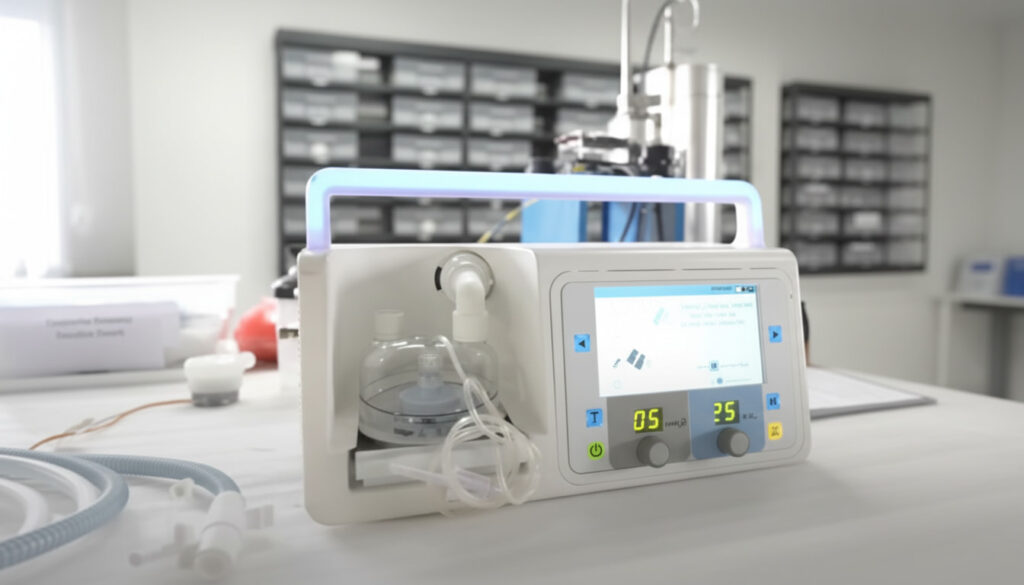
End-to-end redesign and scalable manufacturing transformation of a neonatal CPAP device delivering reliable respiratory support for neonates with Respiratory Distress Syndrome (RDS) – optimized for cost reduction without compromising safety or clinical performance.
Key Impact Metrics
- ~40% BOM cost reduction through systematic engineering optimization
- 30% overall device cost reduction via DFM-led redesign
- Enhanced reliability validated through MTBF simulations and lifecycle testing
- LMIC-ready architecture enabling broader global accessibility
Discuss Your Critical Care Device Program
Client Overview
The partner sought to develop and manufacture a cost-efficient, reliable, and user-centric CPAP system for neonatal Respiratory Distress Syndrome (RDS). The primary objective was to achieve significant cost reduction while preserving clinical performance, safety, and regulatory compliance, thereby expanding access to high-quality respiratory care in low- and middle-income countries (LMICs).
Business Challenge
Strategic Challenge
Meet growing global demand for affordable neonatal respiratory support by achieving substantial cost reduction without diluting product quality or compromising clinical outcomes.
Technical Challenge
The existing system architecture was over-engineered, increasing complexity and cost. Usability constraints affected clinical efficiency, and limited serviceability contributed to downtime. A comprehensive redesign across mechanical, electronic, and usability layers was required to balance safety, performance, and affordability.
Regulatory & Compliance Challenge
The device required alignment with international compliance standards, including structured compliance testing, CDSCO licensing, and CE marking. All design modifications were executed to maintain regulatory integrity while enhancing reliability and optimizing cost structure.
Manufacturing & Scale Challenge
Scalable manufacturing required design simplification, modular architecture, and streamlined assembly processes. Key cost drivers within the BOM required identification and optimization while maintaining predictable production quality.
Our Approach
Comprehensive BOM Teardown & Cost Driver Analysis
A structured BOM teardown identified high-cost components and system inefficiencies. This analysis informed targeted redesign interventions across mechanical and electronic subsystems.
Design for Manufacturing (DFM) Optimization
- Standardized components to reduce variability
- Simplified assembly workflows to minimize production time
- Modular re-engineering to improve field serviceability
- Architecture rationalization to eliminate unnecessary complexity
Industrial & Mechanical Design Optimization
Device geometry and structural elements were optimized to reduce material consumption and enhance manufacturability. Ergonomic refinements supported intuitive clinical handling, and accessibility improvements streamlined servicing and maintenance workflows.
Electronics & Electrical Design Optimization
- Replacement of high-cost PCBs, blowers, LCDs, and battery components with cost-effective alternatives
- Simplified circuitry and reduced component count
- Streamlined electronics architecture supporting intuitive operation
Reliability Engineering
A tool-driven MTBF simulation framework was implemented and validated through lifecycle testing to ensure device durability and consistent performance under operational stress.
Regulatory Enablement
Compliance testing, CDSCO licensing facilitation, and CE marking alignment were integrated with the design transformation process to ensure regulatory continuity.
Results & Business Impact
Measurable Outcomes
- ~40% BOM cost reduction
- 30% overall device cost reduction
- Improved reliability validated through MTBF simulation and lifecycle testing
Operational Impact
- Simplified assembly processes supporting scalable production
- Reduced component complexity to accelerate manufacturing cycles
- Modular architecture supporting faster field repair and minimized downtime
Regulatory Outcome
- Successful compliance testing alignment
- CE marking and CDSCO licensing facilitation
Market Outcome
- Cost-optimized and ergonomic neonatal CPAP solution
- Engineered for deployment in low- and middle-income countries
- Enhanced global accessibility of neonatal respiratory care
Engineer Critical Care Devices for Cost Efficiency, Compliance, and Global Accessibility
As a design-led medical device CDMO, we integrate engineering optimization, regulatory alignment, and scalable manufacturing to transform complex critical care technologies into commercially sustainable and globally accessible solutions.