XEL RF
Jump-start your radiofrequency device development with a proven, multi-application technology platform.

A Faster Starting Point for Your RF Innovation
Radiofrequency (RF) devices often require long development cycles, requiring repeat rounds of verification for every change in modality, probe, or waveform.
Syrma Johari MedTech’s XEL RF offers a pre-validated technology architecture that gives medical device manufacturers a stronger starting point when developing medical-grade RF systems.
XEL RF is built on our deep expertise and hands-on experience in designing and developing RF systems for aesthetic procedures, rehabilitation, and therapeutic applications. Over the past four decades, we have helped our customers in refining product designs and aligning with regulatory standards early to successfully bring RF-based innovations to global markets.
XEL RF gives your RF device a validated base platform that supports common clinical pathways:
Pre-qualified RF hardware modules
Pre-tested firmware with configurable power, waveform, and safety parameters
A flexible interface layer that can be adapted for product-specific UI and control logic
Regulatory documentation templates to streamline submissions for international markets (including the U.S. and EU)
How XEL RF Enables Faster Go-to-Market
Through a series of forward-thinking initiatives, the company integrates environmentally responsible practices across every facet of its business. From harnessing renewable energy to reducing waste, SJML is setting new standards in sustainable manufacturing.
Reduced Design Time

Rapid prototyping with configurable hardware, firmware, and interface layers.
Reduced Engineering Rework

Faster iteration when developing product families or system variants.
Reduced Regulatory Risk

Regulatory documentation and compliance activities progress in parallel with product development.
What You Get With XEL RF
Accelerate your medical device innovation with a proven, compliant foundation.

40–50%
Faster Development
Accelerate cycles by leveraging validated reference architecture.

Up to 25%
Lower NRE Cost
Reduce non-recurring engineering costs through reuse of tested modules.

Reduced
Rework & Redesigns
Minimize engineering rework linked to performance and regulatory compliance.
Qualified
Supplier Ecosystem
Minimize risk and support long-term scalability with trusted partners.

Smoother
Regulatory Path
Streamline FDA 510(k) and EU MDR approvals with pre-validated components.
Ready to Accelerate?
Start your project with the XEL-RF advantage today.
Adaptable Across a Wide Range of RF Devices
XEL RF can be customized for usage across diverse RF devices for medical aesthetics, rehabilitation, and therapeutic applications.

RF and Microneedling Radiofrequency (MNRF)-Based Medical Aesthetics
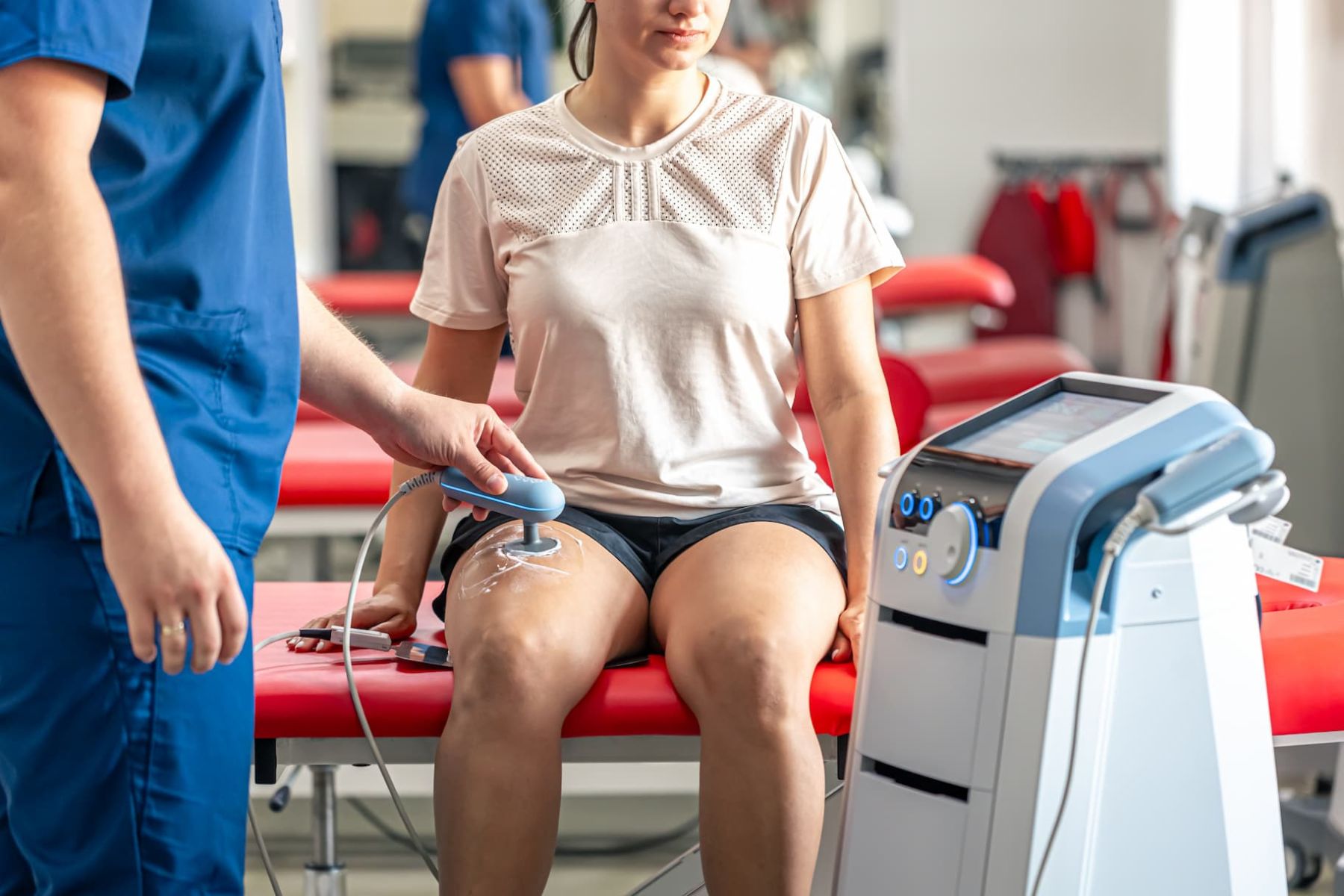
Physiotherapy and Rehabilitation
RF Ablation and Electrosurgery
A Few of Our Customers’ Devices Brought to Life Using XEL RF

Precision Microneedling RF Platform for Facial Rejuvenation

Solid-State Diathermy System for Physical Medicine and Rehabilitation

Class II, 510(k)-cleared monopolar RF body contouring Device
Drive your RF product closer to commercialization with a reliable technology foundation.
Key Questions Answered
1. What is XEL RF?
XEL RF is a pre-validated technology platform to speed up the development of medical-grade radiofrequency devices. It provides a proven hardware, firmware, and system architecture that helps reduce development time and engineering rework when developing RF-based products.
2. Who is XEL RF intended for?
XEL RF is intended for medical device companies launching RF systems for medical aesthetics, rehabilitation, and therapeutic applications. It is particularly suited for teams looking to shorten development cycles while maintaining performance and regulatory readiness.
3. How does XEL RF help reduce development timelines?
By offering pre-qualified RF hardware modules, pre-tested firmware, and reusable system architecture, XEL RF allows teams to avoid rebuilding core RF subsystems. This enables faster prototyping, quicker iterations, and parallel progress on engineering and regulatory activities.
4. Does XEL RF support customization and product differentiation?
Yes. While XEL RF provides a validated base architecture, it supports customization with regard to power levels, waveforms, probes, applicators, UI workflows, and clinical use cases. This allows innovators to differentiate their products while leveraging a common foundation.
5. What types of RF devices can be developed using XEL RF?
XEL RF can support a wide spectrum of RF devices, including microneedling RF platforms, solid-state diathermy systems, monopolar and bipolar RF body contouring devices, and other therapeutic and rehabilitation-focused RF systems.
6. How does XEL RF support regulatory compliance?
XEL RF is developed with regulatory requirements in mind. The platform includes reusable documentation templates and design considerations compliant with global standards. It helps streamline regulatory submissions for markets including the U.S. (FDA 510(k)) and the EU (MDR).
7. How can we evaluate whether XEL RF fits our next RF device project?
Evaluation typically begins with a discussion to understand your application, clinical requirements, and target markets. From there, relevant elements of the XEL RF platform can be mapped to your product roadmap to assess fit and reuse potential.
Have a Question?
If you have a specific question or would like more information about XEL RF, submit your query and our team will get back to you shortly.
