Leading MDSAP & FDA-Certified Medical Device CDMO
Contact Syrma Johari MedTech
From Design to Delivery: Accelerate Your Time-to-Market with Regulatory Excellence
Our regulatory experts respond within 24-hours





Why Choose Syrma Johari MedTech?
Speed to Market
Reduce development time by 30% with our integrated Design, Manufacturing, and QARA services. From prototype to production in record time.
Regulatory Excellence
MDSAP audited, FDA GMP compliant, ISO 13485 certified. Our QARA experts navigate complex global approval pathways for FDA (US), CE (EU), PMDA (Japan), and NMPA (China).
Cost Optimization
Scale production efficiently with our state-of-the-art manufacturing facility. Reduce per-unit costs while maintaining premium quality standards.
Our Success Stories




Who We Are?
We are a leading Indian manufacturer of electronic medical devices, partnering with global companies to design, develop, and produce a wide range of healthcare products
Years Experience
Exporting Countries
Clients
Awards & Recognitions
Our Solutions
Design & Engineering
Mauris blandit aliquet elit, eget tincidunt nibh pulvinar a. Vestibulum ac diam sit
Contract Manufacturing
Mauris blandit aliquet elit, eget tincidunt nibh pulvinar a. Vestibulum ac diam sit
PCB Assembly
Mauris blandit aliquet elit, eget tincidunt nibh pulvinar a. Vestibulum ac diam sit
Full Medical Device Lifecycle
Mauris blandit aliquet elit, eget tincidunt nibh pulvinar a. Vestibulum ac diam sit
Quality Assurance & Regulatory Affairs Services
Mauris blandit aliquet elit, eget tincidunt nibh pulvinar a. Vestibulum ac diam sit
Explore All Services →
Mauris blandit aliquet elit, eget tincidunt nibh pulvinar a. Vestibulum ac diam sit
Get Started
Ready to Accelerate Your Medical Device?
Prefer to talk now?
Phone (India): +91-1800-102-8684 (Toll-free)
Phone (US): +1-918-918-3277 (Miami office)
Call our regulatory experts directly for immediate guidance.
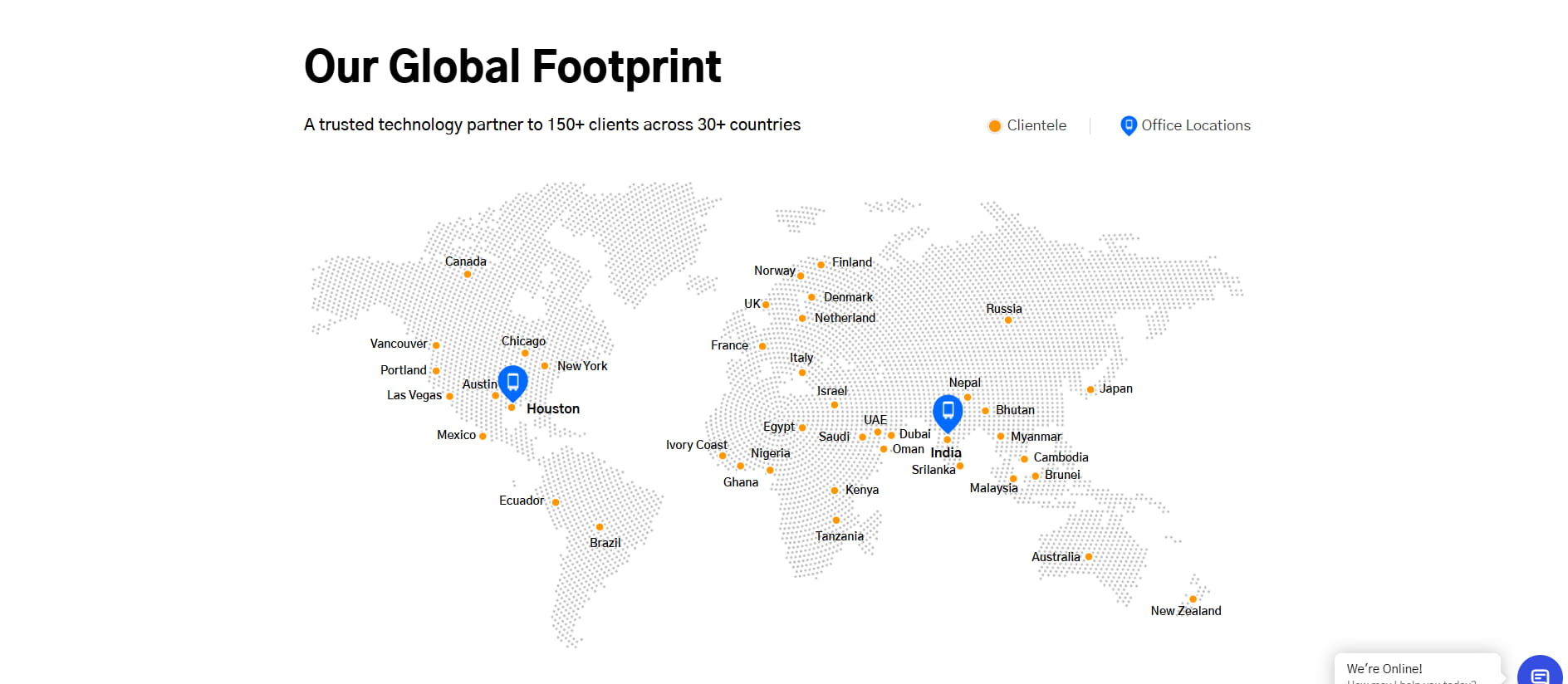
Our Office Locations
Meet us in person at our offices in the USA and India

Jodhpur, India

Pune, India
Design & Innovation Center
World Trade Center, 402, Tower 2,
WTC, Kharadi, Pune-411014, India
Frequently Asked Questions
How long does it take to get FDA approval?
Timeline depends on device classification and complexity. With our QARA support, most 510(k) submissions are completed within 6-9 months. High-risk PMA devices typically require 2-3 years. We provide a detailed regulatory timeline during your initial consultation.
What's the minimum order quantity for contract manufacturing?
We support projects from low-volume prototyping (10-100 units) through mass production. Our flexible manufacturing model scales with your business needs. No minimum order quantity required for initial production runs.
Do you work with international companies?
Yes! We serve OEMs from 30+ countries. We support FDA (US), CE marking (EU), PMDA (Japan), NMPA (China), and other regulatory pathways. Our team is experienced with multi-market product launches.
What certifications do you hold?
We maintain ISO 13485:2016, ISO 27001:2022, MDSAP certification, and US FDA GMP compliance. Our facilities and processes undergo regular audits to maintain these certifications.
How quickly can you respond to inquiries?
Our target is to respond within 24 business hours for all inquiries. For urgent requests, call us directly at +91-1800-102-8684 (India) or +1-918-918-3277 (US).

